In the previous note from the chairman I emphasized the potential role for Ten Bio in the area of wound management. My reason for that emphasis is simple: if you are evaluating how well a new approach / treatment can influence wound healing, then the key attribute of the test platform must be that it exhibits human skin’s normal response to wounding in vivo . . . or as close to ‘normal’ as can be achieved!
Wound healing is a complex, multifaceted process which requires a coordinated set of actions from multiple cell types and extracellular components which exist within all layers of the skin. It is only when this harmony is achieved that the body can achieve the repair that we observe – haemostasis, inflammation, proliferation, re-epithelialisation and remodelling.
Therefore the test platform must demonstrate two key attributes:
- it must maintain the integrity of the skin layers with the full cellular profile
- the platform should mimic the mechano-biology of the skin in vivo to ensure that the complex process of repair can take place.
Testing a treatment innovation on a platform that does not have these attributes can only deliver incomplete and potentially misleading results.
Testing to date has relied upon a range of platforms – in vivo animal models, 3D skin models constructed in the lab, and ex vivo skin models. All have their limitations in assessing new wound healing approaches. Animal models show significant variation across species, and their differences to human skin hinder their application in skin research. Lab grown tissue lacks the full range of anatomical features and cell types, and ex vivo models lose integrity and viability quickly.
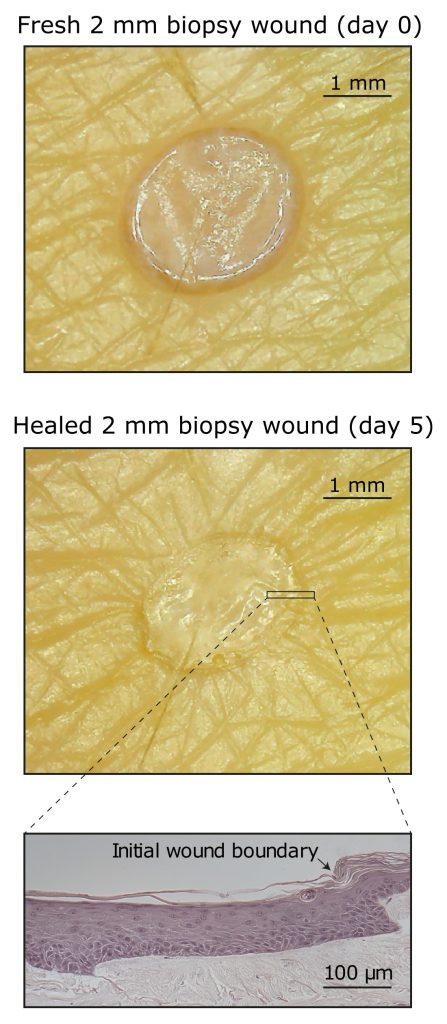
Ten Bio’s model, TenSkin™, is full thickness skin held at an optimal tension which confers on the model the ability to closely mimic skin’s behaviours in vivo and for the model to be viable for a much longer time – up to 14 days.
In our testing we have noted that 2 mm wounds fully close and heal without any external interference. We have also shown that the process by which the wound heals is very similar to that seen in equivalent wounds in humans in clinical assessments.
Our team is continuing to explore the capabilities of the TenSkin™ brand in wound healing and other test situations. Our data base on the performance of TenSkin™ in a range of assaults or treatments is growing rapidly. As is our experience and data related to the performance of skin across the full range of the Fitzpatrick scale.
The longer duration of TenSkin™’s viability and its mimicking of human skin’s in vivo performance is delivering excellent value to Ten Bio’s clients in industry, academia, and the public sector.
TenSkin™ and the capabilities of the Ten Bio team provide to our clients a reliable baseline of skin’s performance, which enables a wide variety of testing. The outcome of this testing is better data that is more predictive of performance in the clinic. Improved quality of data and its analysis leads to better decision making as our clients pursue the answers to their development questions. Cost savings are a welcome accompaniment to this improved quality of research tools and techniques.
The ability to deliver that value into the high risk process of new product development is a major plus for Ten Bio and its products and services.
Why use anything else?
Ken Fyvie
02.08.22



